Most of the studies performed with broadband spectroscopy have been performed with Fourier-Transform Infrared Spectrometers (FTIR), which are equipped with thermal light sources. However, the moderate power of the thermal light source and the low sensitivity of available mid-infrared detectors limit the achievable sensitivity. This problem is further aggravated for liquid or aqueous samples. Water is a strong absorber in the mid-infrared (MIR) wavelength range. Therefore, in the majority of cases the thickness of the sample is usually limited to ≈10 µm. Besides limiting the applicability to very thin samples, practical problems arise, like clogging of liquid cuvettes.
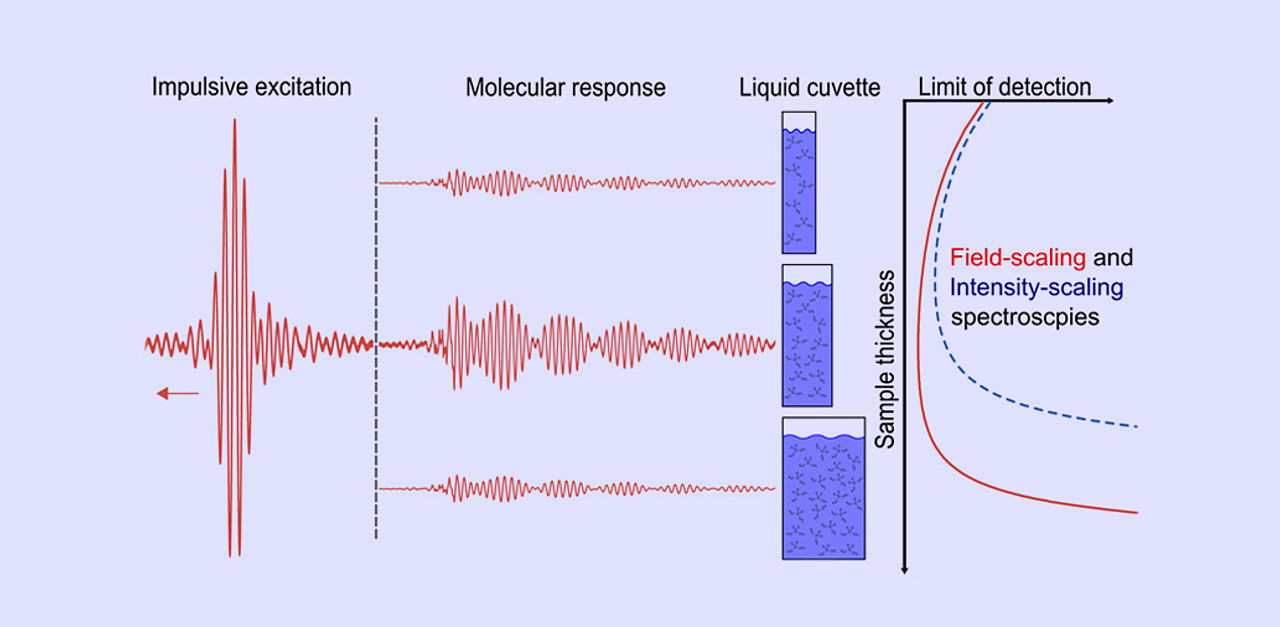
In a recent paper, our team has demonstrated in theory and experiment the potential of field-resolved infrared spectroscopy (FRS) to overcome these long-standing limitations. FRS relies on the excitation of resonant molecular vibrations with waveform-stable, broadband MIR pulses, and electric-field-resolved detection of the emerging fingerprint waveforms. FRS makes it possible to record a molecular signal with high signal-to-noise ratio, although water attenuates the excitation by orders of magnitude. We found that sub-µg/ml detection sensitivities can be maintained for samples as thick as 80 µm and that even for 0.2-mm-thick samples, detection sensitivities in the range of 10 µg/ml are feasible, which is the level achieved by state-of-the-art research-grade FTIR instruments, albeit under the stringent condition of sub-10-µm sample thickness.
The relaxed requirements to the sample thickness enable a more flexible design of liquid cuvettes and microfluidic chips for MIR spectroscopic applications. In addition, the increased signal-to-noise ratio for the measurement of thick aqueous samples will benefit MIR transmission spectroscopy and spectro-microscopy of biological samples, such as living cells, bulk cell and tissue-cultures as well as biological tissues, in their natural (hydrated) state.